NEW FINDINGS ON THE STRUCTURE OF THE FOXO4: p53 COMPLEX - A KEY FACTOR IN SENESCENCE REGULATION (9.4.2022)
Transcription factor p53 protects cells against tumorigenesis when subjected to various cellular stresses. Under stress conditions, p53 interacts with another transcription factor, FOXO4 (Forkhead box O 4), and together they increase the production of p21 protein, which triggers the process of cell aging (senescence). However, the molecular mechanism of upregulation of p21 transcription is still unclear. In a work published in the Protein Science journal, scientific teams of Dr. Obsilova (IPHYS CAS), prof. Obsil (Faculty of Science, Charles University and IPHYS CAS) and their colleagues from IOCB CAS characterized interactions between p53 and FOXO4 at the molecular level. New knowledge about the structure of the complex may enable the development of specific inhibitors of the interaction between these two proteins, and subsequently in the development of new drugs aimed at the selective elimination of senescent cells.
In this structural study, the researchers performed a detailed characterization of the interactions in the FOXO4: p53 complex using an integrated approach involving analytical ultracentrifugation, nuclear magnetic resonance, and chemical cross-linking coupled to mass spectrometry. Because both FOXO4 and p53 have multiple domains (see Figure), they studied the role of individual domains and disordered segments of both proteins and mapped their interaction interfaces. They found out that the interaction between p53 transactivation domain TAD and the FOXO4 Forkhead domain is crucial for the overall stability of the p53:FOXO4 complex. Furthermore, contacts involving the N-terminal disordered FOXO4 segment, the C-terminal negative regulatory domain of p53, and the DNA-binding domains of both proteins stabilize the complex formation. By measuring DNA binding, they further found that the p53: FOXO4 complex formation blocks p53 binding to DNA without affecting the DNA-binding properties of FOXO4.
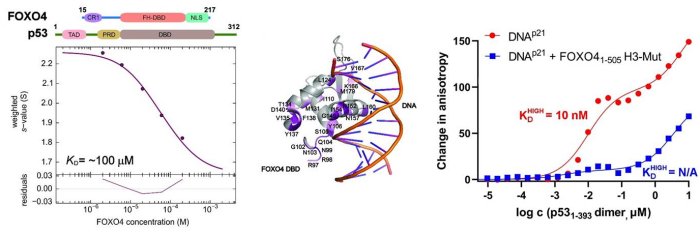
Left, sedimentation velocity analytical ultracentrifugation analysis of interaction between FOXO4 and p53. Middle, chemical shift perturbations obtained from 1H-15N HSQC spectra of 15N-labeled FOXO4 in the presence of p53 mapped onto the crystal structure of the FOXO4 DBD:DNA complex. Right, fluorescence anisotropy measurements showing that the complex formation reduces the DNA-binding affinity of p53.
Mandal R, Kohoutova K, Petrvalska O, Horvath M, Srb P, Veverka V, Obsilova V and Obsil T. FOXO4 interacts with p53 TAD and CRD and inhibits its binding to DNA. Protein Sci. roč. 31, č. 5 (2022), č. článku e4287. IF = 6.725. DOI: 10.1002/pro.4287.
