Sensor domain of TRPA1 channel contributes to detection of painful stimuli
Acute pain is a normal reaction of the somatosensory system to noxious stimulation which serves a protective function and is the first symptom of disease, especially of inflammatory origin. In its chronic form, pain is maladaptive and has a significant impact on quality of life. The scientists from the Department of Cellular Neurophysiology, Institute of Physiology of the Czech Academy of Sciences, in collaboration with the Division of Biomolecular Physics at the Faculty of Mathematics and Physics, Charles University, investigate the function of a distinct group of proteins involved in the mechanisms of acute and chronic pain.
The survival of all living organisms is determined by the ability to promptly recognize potentially harmful or painful stimuli. In mammals, this „alarm system“ constitutes of specialized neurons called nociceptors. On the nociceptors, several members of a distinct group of proteins called transient receptor potential (TRP) are expressed. These proteins form ion channels that can detect painful stimuli of various qualities (chemical, thermal or mechanical) and transduce the information into an electrical signal. The ankyrin receptor TRPA1 is one of the TRP ion channels, that can be activated by endogenous pain-causing substances. Temperature, pressure, various environmental irritants, pungent natural compounds from cinnamon or mustard oil, and other molecules generated by injury or inflammation activate TRPA1 in an allosteric manner, meaning that the channel’s response to one stimulus alters its response to another. In the latest study, the scientists from the Institute of Physiology of the Czech Academy of Sciences in collaboration with the experts from the Faculty of Mathematics and Physics, Charles University, investigated the structure of TRPA1 and found an intracellular region within the sensor domain forming a cavity that coordinates this complex allostery. For example, mutations in the cavity prevented stabilization of the channel by interacting with intracellular phospholipids, resulting in dysregulated sensitivity to calcium ions or membrane voltage. These findings contribute to the understanding of molecular mechanisms by which TRPA1 channels are gated and how these mechanisms help the channels to play their physiological parts in chronic pain.
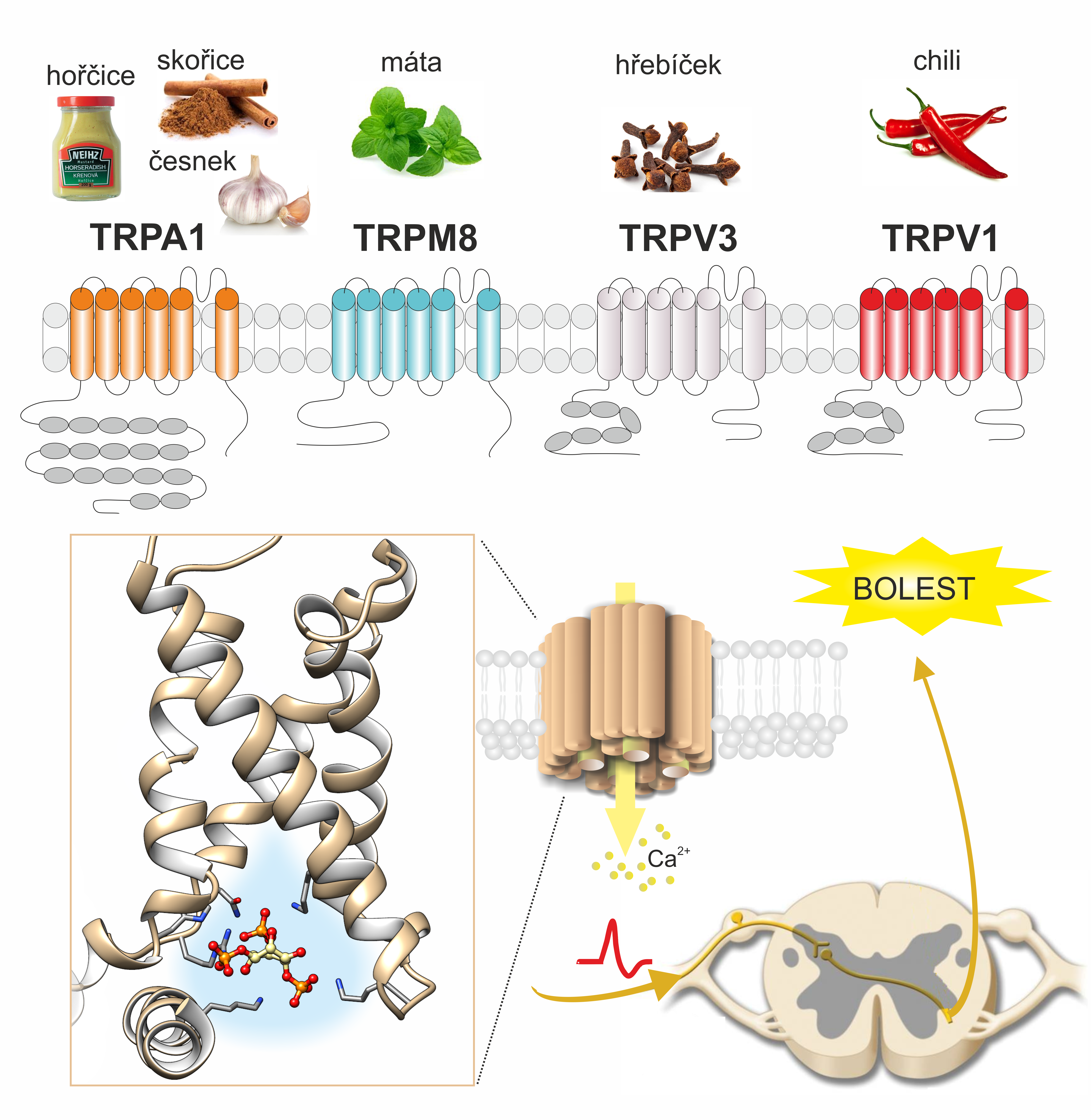
Citace: L. Zimova, V. Sinica, A. Kadkova, L. Vyklicka, V. Zima, I. Barvik, V. Vlachova, Intracellular cavity of sensor domain controls allosteric gating of TRPA1 channel. Science Signaling, 2018, vol. 11, Issue 514, DOI: 10.1126/scisignal.aan8621
