Targeted modulation of NMDA receptors is a key for the effective treatment of neurological and neurodevelopmental diseases
N-methyl-D-aspartate (NMDA) receptors are glutamate-gated ion channels critically involved in excitatory synaptic transmission that play a key role in learning and memory. Impaired NMDA receptor function leads to major neurological, neurodevelopmental and psychiatric disorders such as schizophrenia, autistic spectrum disorder, epilepsy or Alzheimer's disease. For an effective design of novel drugs capable of specifically modulating NMDA receptors, it is essential not only to understand the NMDA receptor atomic structure, but also to uncover the specific sequence of conformational changes that are involved in receptor activation and allosteric modulation.
We used single-molecule FRET to identify and quantify the sequence of conformational changes in the amino-terminal domain of the NMDA receptor during its activation. Next, we uncovered distinct roles of receptor subunits in receptor activation, and last but not least, we have identified the molecular mechanism of receptor modulation by pH during pathophysiological conditions such as stroke.
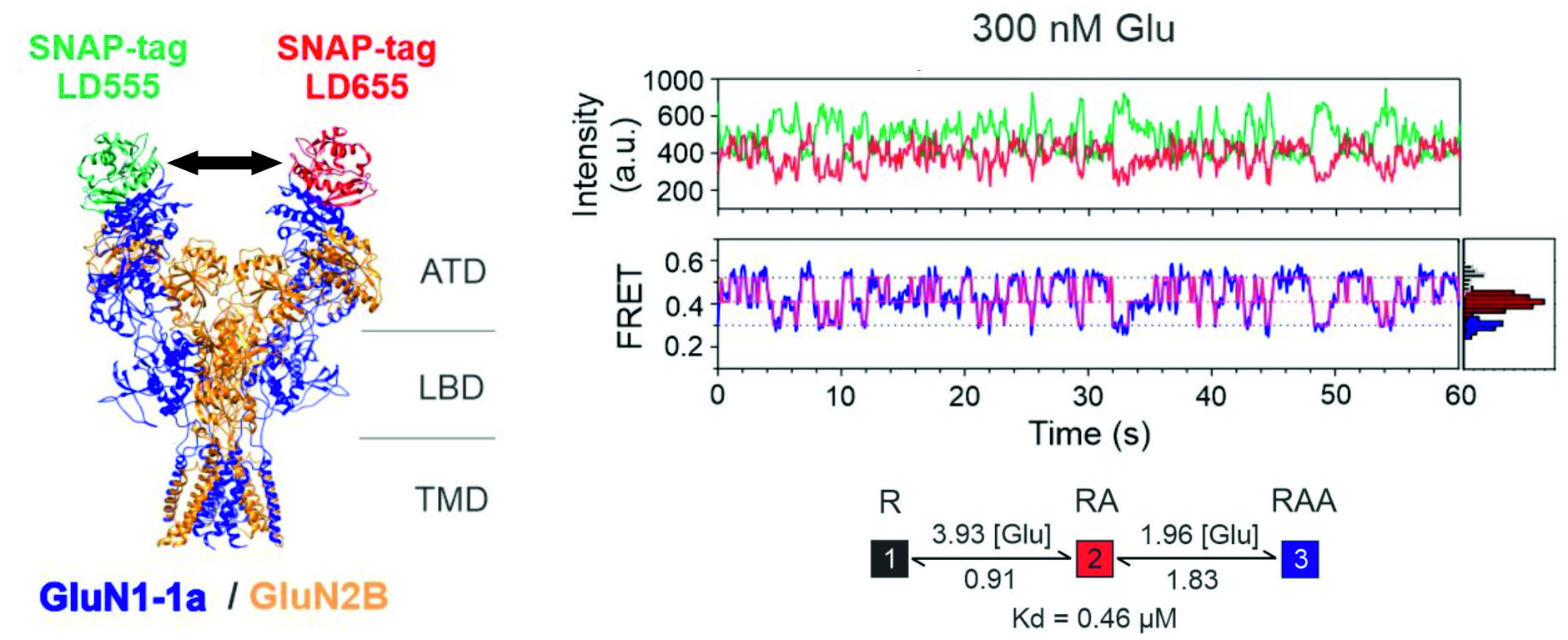
Vyklický, Vojtěch - Stanley, Ch. - Habrian, Ch. - Isacoff, E. Y. Conformational rearrangement of the NMDA receptor amino-terminal domain during activation and allosteric modulation. Nature Communications. Roč. 12, č. 1 (2021), č. článku 2694. ISSN 2041-1723. E-ISSN 2041-1723, IF: 14.919, rok: 2020, DOI
