Metabolomika, lipidomika a fluxomika
| Software: |  GTTAtlas GTTAtlas |
 MetaboAtlas21 MetaboAtlas21 |
 Mapper Mapper |
 Reaction network viewer Reaction network viewer |
 LORA LORA |
a výběr speciálních metod z oblasti metabolomiky, lipidomiky a fluxomiky.
 GTTAtlas
GTTAtlas
metabolomický atlas myších tkání během orálního glukózového tolerančního testu (OGTT) s využitím [13C6]-glukózy
- Interaktivní metabolomický, lipidomický a fluxomický atlas polárních a nepolárních metabolitů.
- Standardní orální glukózový toleranční test.
- Dvanáct metabolicky relevantních orgánů a plasma bylo analyzováno v časovém profilu OGTT.
- Šestidimenzionální LC-MS platforma byla použita na analýzu vzorků.
- Hmostnostní spektrometrie byla použita na detekci metabolitů značených 13C.
- Osud 13C-značené glokózy byl mapován na metabolické dráhy.
- Metabolické vazby uvnitř a mezi orgány byly vyhodnoceny pomocí pokročilých statistických metod.
- Software pro mapování analytů na metabolické dráhy Mapper - a pathway mapping tool.
► Magno Lopes, Kristyna Brejchova, Martin Riecan, Michaela Novakova, Martin Rossmeisl, Tomas Cajka, Ondrej Kuda✉
Metabolomics atlas of oral 13C-glucose tolerance test in mice
Cell Reports, 2021, 27 (2), 109833 DOI https://doi.org/10.1016/j.celrep.2021.109833
- A systems-level organ-specific metabolomics atlas of mouse tissues
- 13C6-glucose tracer tissue distribution during glucose tolerance test
- Interactive online application for metabolic pathway exploration
- LC-MS data resource covering polar metabolites and complex lipids
 MetaboAtlas21
MetaboAtlas21
MetaboAtlas21 je zevrubný atlas myšího metabolomu a lipidomu.
- Ve spolupráci s Laboratoří metabolomiky.
- Computational resources were supplied by the project "e-Infrastruktura CZ" (e-INFRA LM2018140) provided within the program Projects of Large Research, Development and Innovations Infrastructures.
 Mapper - a pathway mapping tool
Mapper - a pathway mapping tool
- Online nástroj pro mapování metabolitů na metabolické mapy.
- Pomocí excelového vstupního souboru lze vizualizovat hladiny metabolitů.
- Podporuje jednoduché sloupcové grafy i složené grafy pro isotopology.
 Metabolic model viewer
Metabolic model viewer
- Online nástroj pro vizualizaci metabolických modelů.
- Podporuje pouze 13C-značené experimenty.
- Kompatibilní s MFA Suite™ (Metabolic Flux Analysis Suite) https://mfa.vueinnovations.com/.
 LORA
LORA
- Online nástroj pro Lipid Over-Representation Analysis.
- Postaven s použitím softwaru Goslin, díky čemuž podporuje různé lipidové názvosloví.
- Používá UpSet plot, circulární hierarchický strom and funkce Cytoscape network k popsání obohacení lipidomu.
► Michaela Vondrackova, Dominik Kopczynski, Nils Hoffmann, Ondrej Kuda✉
LORA, Lipid Over-Representation Analysis Based on Structural Information
Analytical Chemistry, 2023 DOI https://doi.org/10.1021/acs.analchem.3c02039
- Webová aplikace LORA.metabolomics.fgu.cas.cz
- GIT https://github.com/IPHYS-Bioinformatics/LORA
Dynamická metabolomika - stopování osudu 13C glukózy
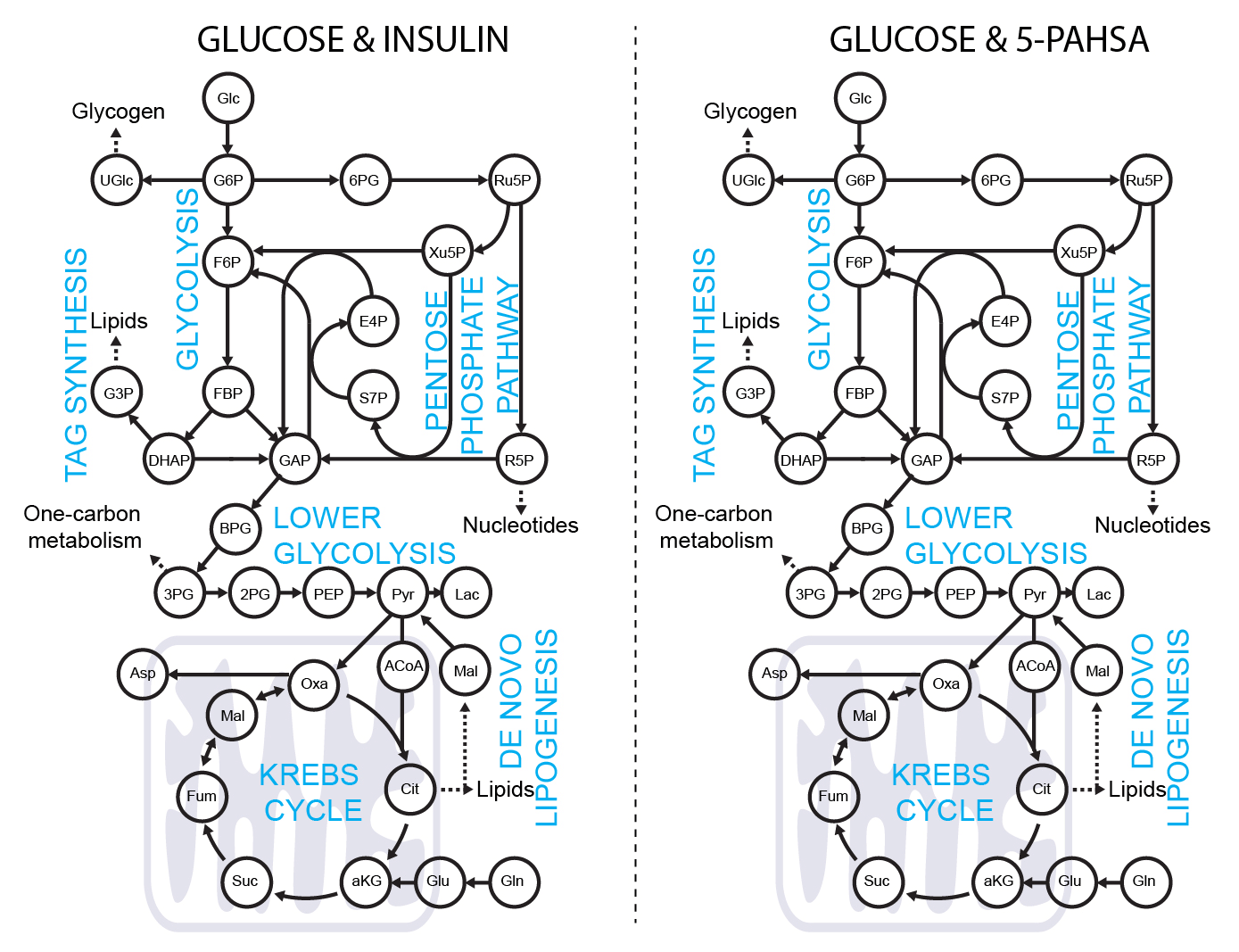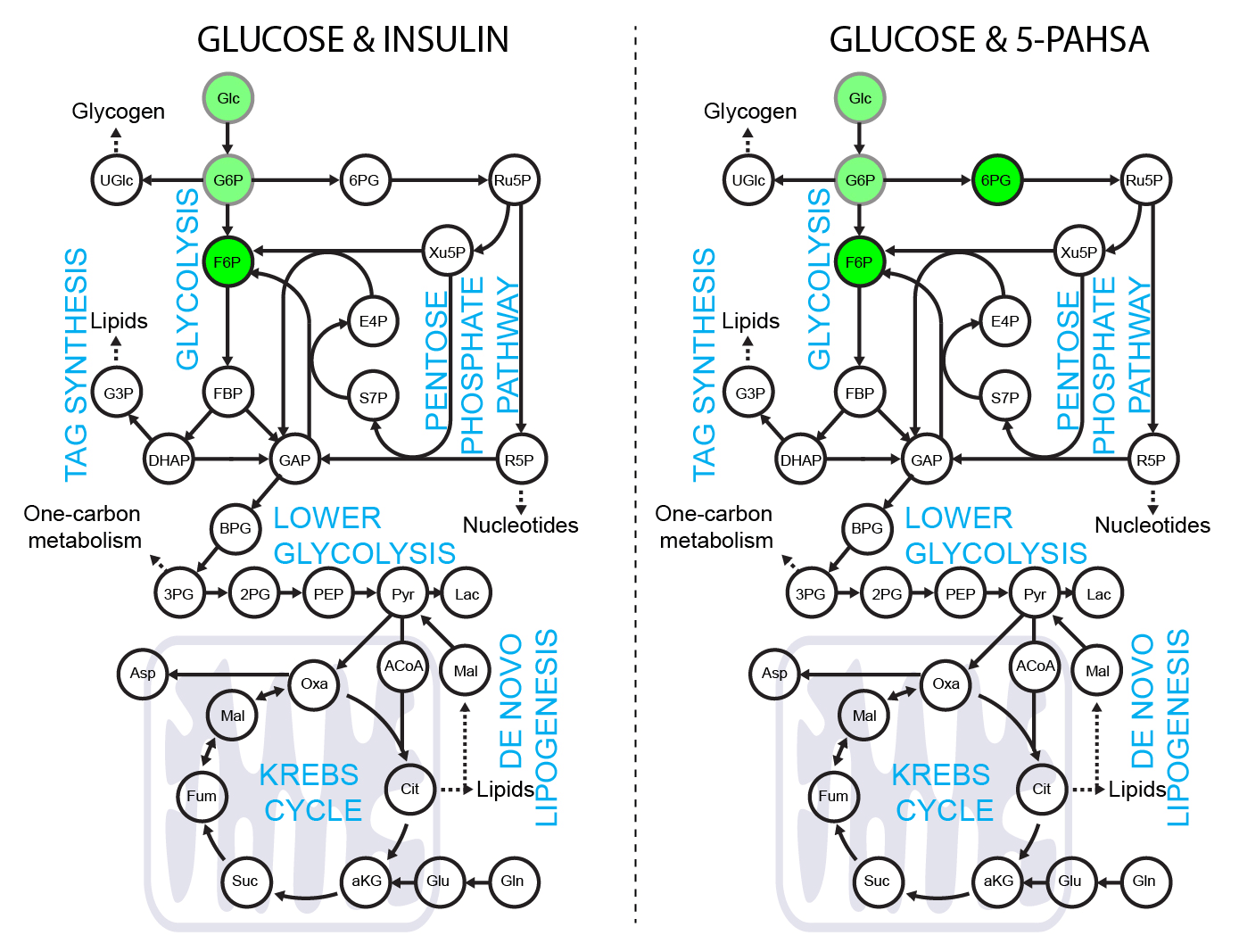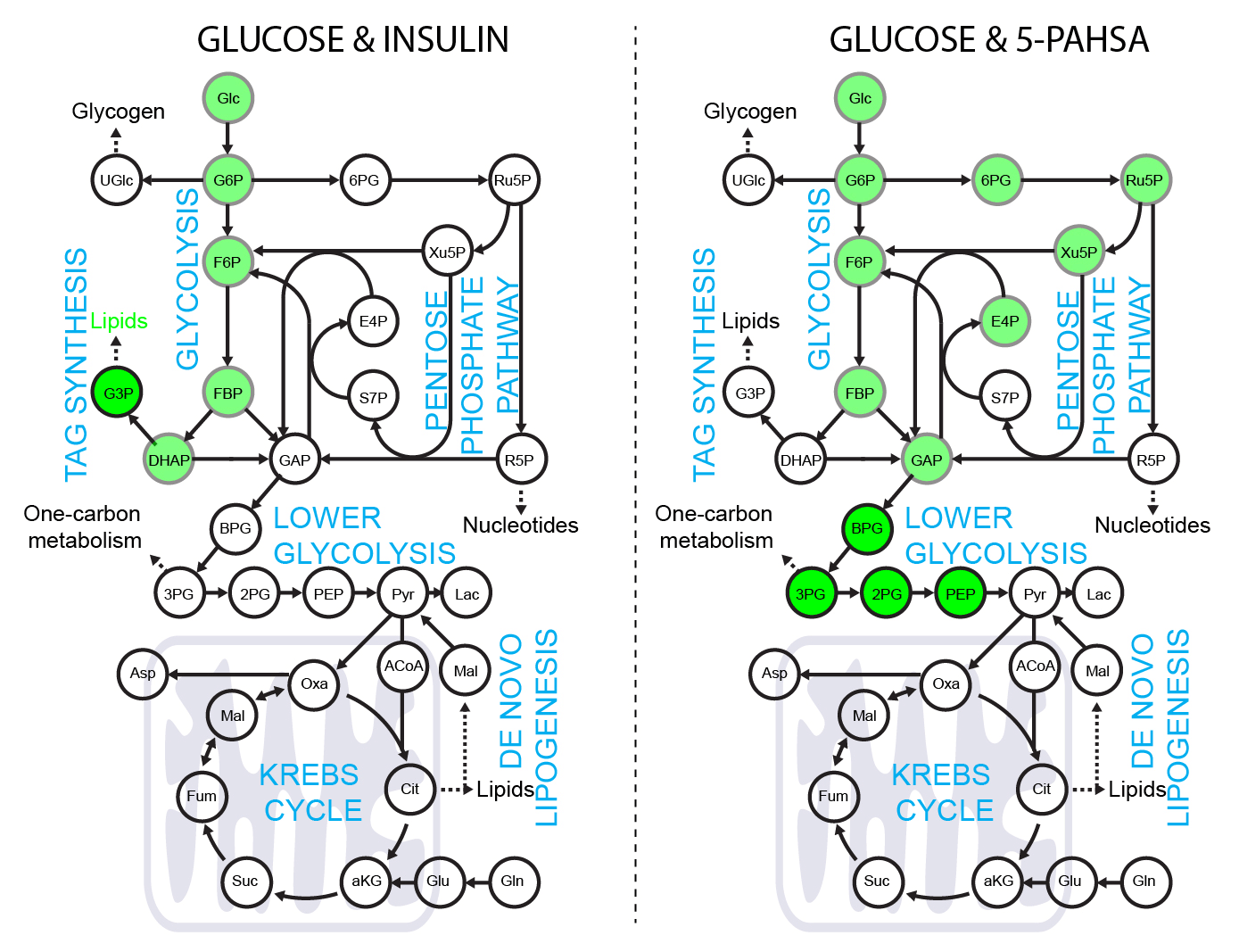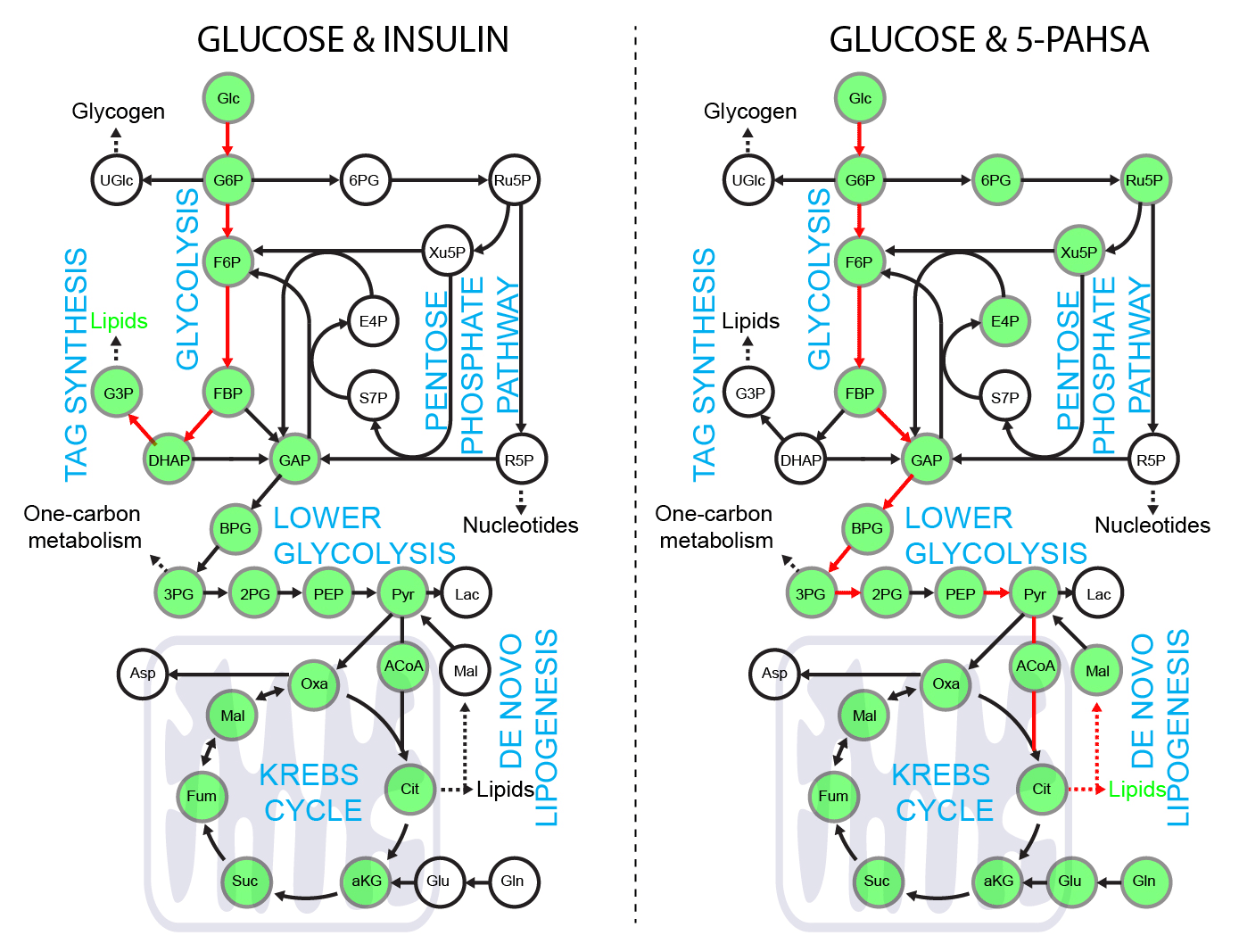
5-PAHSA primes adipocytes for glucose metabolism in a different way from insulin. 5-PAHSA partially re-wires glucose metabolism pathways in favor of Krebs cycle, NADPH synthesis and de novo lipogenesis. Simplified metabolic situation is animated through the pathway within 15 minutes of glucose uptake into adipocytes. Details and conditions in Paluchova et al. DOI and FAHFA page
Použití [4-2H]-glukozy k metabolickému značení NADPH.
[4-2H]-glucose can be used as a malic enzyme tracer, which specifically labels NADPH’s redox active hydrogen. The deuteron from [4-2H]-glucose is passed to NAD2H by glyceraldehyde 3-phosphate dehydrogenase (GAPDH) during glycolysis. This cytosolic NAD2H can be used by cytosolic malate dehydrogenase 1 (MDH) to convert cytosolic oxaloacetate (coming from citrate cleavage by ATP citrate lyase) to cytosolic [2-2H]-malate. This deuterated malate can be either transported to mitochondria or converted by malic enzyme 1 (ME1) to cytosolic pyruvate and cytosolic NADP2H. This NADP2H represents the energy for DNL. During the synthesis of a fatty acid from acetyl-CoA units (via malonyl-CoA), two NADPH molecules are used per one C2 unit. The fatty acid labeling is the result of two stochastic hydrogen selection processes, with 1/3 of hydrogens coming from water and 2/3 coming from NADPH. If cytosolic NADP2H is present, contribution of malic enzyme 1 NADPH to fatty acid synthesis can be measured.
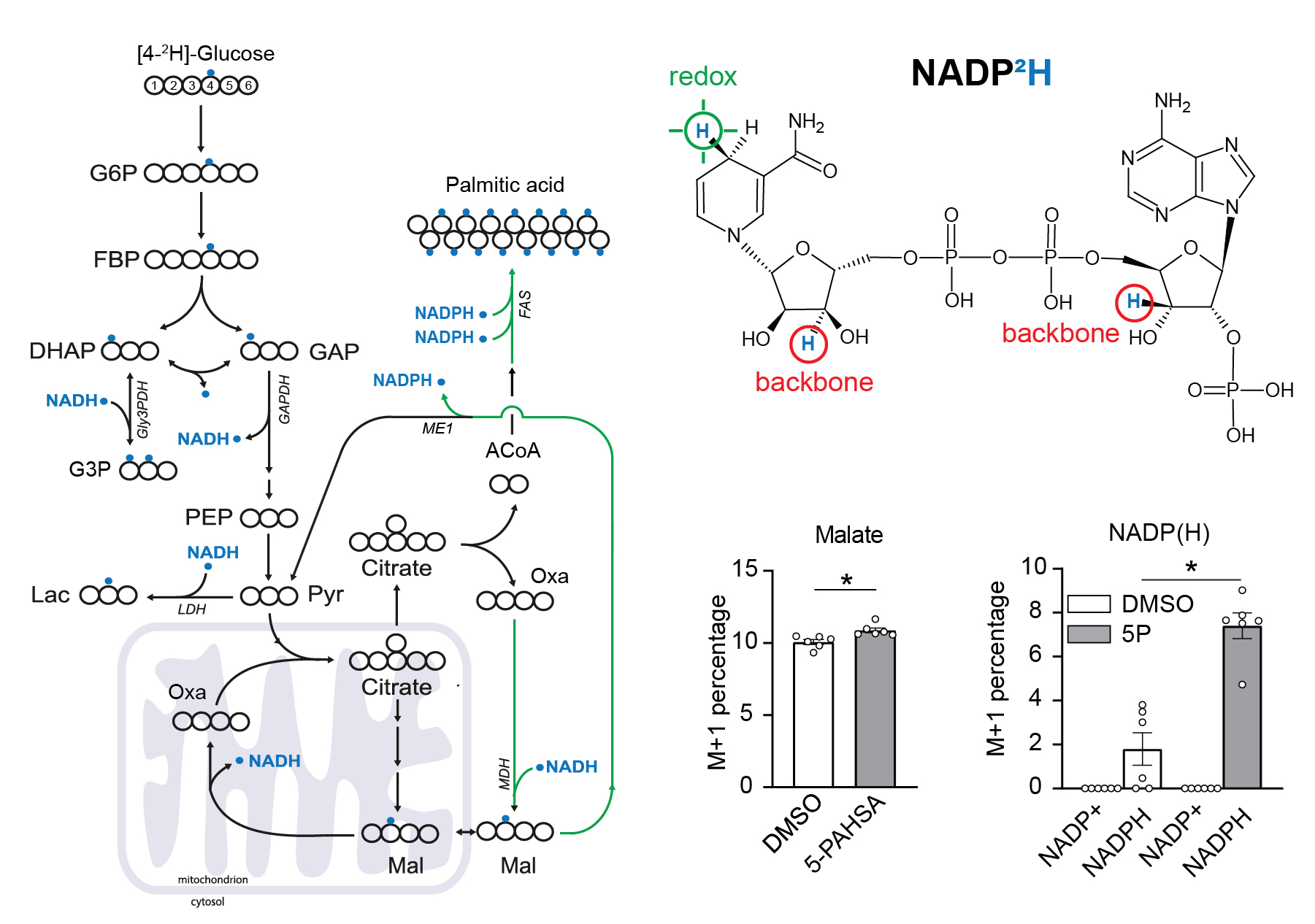
3T3-L1 differentiated adipocytes were incubated with or without 40 µM 5-PAHSA in medium with [4-2H]-glucose for 24 hours without insulin, metabolism quenched on liquid nitrogen, cells extracted to preserve NADPH and NADP+ ratio, and raw extracts immediately measured by LC-MS/MS according to a published protocol.
Malate labeling M+1 documents that the deuteron was introduced by labeled NADH. For further details regarding malate labeling and its alternative fate see ref. Lewis et al. NADPH M+1 over NAPD+ M+1 labeling represent the redox-active deuteron on NADP2H. 5-PAHSA treatment resulted in significantly higher labeling of cytosolic NADPH. For more details see ref. Lie et al. Scheme adapted from Lewis et al.. Data from this labeling experiments were adjusted for C, H, O, N, P natural abundance and tracer purity using IsoCor-2.0.5. Details and conditions in Paluchova et al. DOI and FAHFA page
Značení lipidů pomocí 2H2O - syntéza neutrálních lipidů
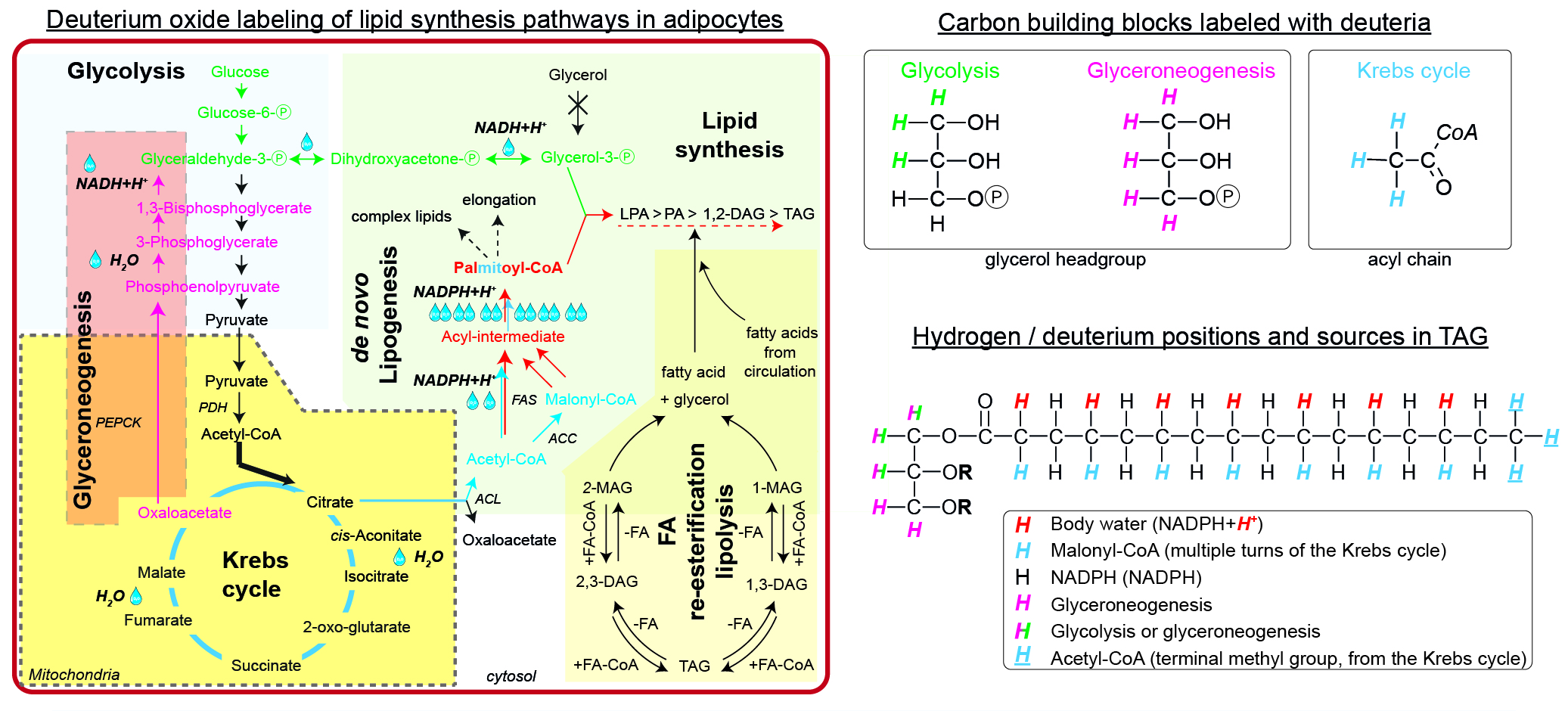
For the simple synthesis of TAGs, glycerol phosphate and acyl-CoAs are needed. WAT is virtually lacking glycerol kinase, thus all glycerol phosphate has to be synthesized either via glycolysis (green path) or glyceroneogenesis (pink path). During glycolysis, deuteria (up to 3, M+3) could be incorporated from water within the equilibrium between Glyceraldehyde-Ⓟ, Dihydroxyacetone-Ⓟ, and Glycerol-3-Ⓟ. The alternative glyceroneogenic pathway precursors are deuterium-labeled within the Krebs cycle and further during conversion of Phosphoenolpyruvate to Glyceraldehyde-Ⓟ. This path may incorporate up to 5 deuteria per glycerol after several turns of the Krebs cycle. The intermediates labeled with deuteria within the Krebs cycle may also enter the de novo lipogenic pathway (blue path) as labeled Acetyl-CoA. The first (and future terminal methyl group) Acetyl-CoA keeps the deuteria, while the later Acetyl-CoAs are converted to Malonyl-CoAs and lose one deuteria from the Krebs cycle. During the elongation process of the acyl-intermediate, NADPH+H+ provides additional deuteria equilibrated from body water, thus potentially increasing the enrichment (red path). Finally, both glycerol phosphate and newly synthesized palmitate can form TAG. The degree of enrichment of the final TAG might be diluted by the contribution of FA re-esterification / lipolysis or import of external fatty acids. There are up to 6 metabolically different hydrogens, which could be exchanged for deuterium in a simple TAG.
