Glutamate receptors
Glutamate is the main excitatory neurotransmitter in the central nervous system. It activates specific membrane proteins known as glutamate receptors. Our studies focus on ionotropic glutamate receptors, which, when activated, allow ions to flow across the cell membrane. The membrane current leads to fast changes in the membrane potential (excitation) and to the spread of information to other neurons. Ionotropic glutamate receptors participate in all neuronal circuits and are thus involved in virtually every function of the nervous system. These receptors mediate changes in synaptic strength, which underlie the formation and storage of memory traces in the brain.
There are three known families of ionotropic glutamate receptors (NMDA, AMPA and kainate receptors), similar in their primary structure, but different in their kinetic properties, sensitivity to different pharmacological agents and membrane distribution. A functional NMDA receptor consists of four GluN subunits. NMDA receptor subunits are coded by 7 genes, with the GluN1 subunit existing in 8 splice variants.
There is much clinical and experimental evidence that despite their essential role in normal physiology, ionotropic glutamate receptors can, under certain pathological conditions, participate in the development of serious psychiatric and neurological disorders. For example, excessive activation of NMDA receptors leads to neuronal death known as excitotoxicty. Excitotoxicity causes brain damage resulting from brain ischemia, perinatal hypoxia, asphyxia, cardiac arrest, and brain trauma. Excitotoxicity is also thought to participate in the loss of neurons and cognitive damage associated with Alzheimer's disease and Parkinson's disease. On the other hand, the hypofunction of NMDA receptors (such as can be triggered pharmacologically) leads to hallucinations similar to those experienced by schizophrenic patients. Experimental evidence indicates that schizophrenia may have genetic causes involving proteins functionally associated with NMDA receptors.
Research Topics
Genetic variants of NMDA receptor genes in schizophrenia patients
Aleš Balík, Ph.D.
Schizophrenia is a psychiatric illness with a significant adverse impact on the patient quality of life. It is known that this disorder involves abnormal glutamatergic signaling. Our goal is to identify changes in the regulatory domains of NMDA receptor genes and/or genetic variants of additional components of the glutamatergic signaling complex in schizophrenia patients. Our experiments aim to characterize the influence of different genetic variants on the function of NMDA receptors and on glutamatergic signaling in general. Our work will help identify possible biomarkers for schizophrenia. (This project is in collaboration with prof. J. Horáček, NUDZ, Klecany.)
Regulation of NMDA receptor function by N-glycosylation
Martin Horák, Ph.D.
While it has been known for over 10 years that NMDA receptors are N-glycosylated, the role of this modification in the regulation of NMDA receptor function is not clear. This project uses a multidisciplinary approach including molecular biology, biochemistry, immunocytochemistry and electrophysiology to determine which N-glycosylation sites and/or which types of N-glycans regulate NMDA receptor transport to the cell membrane and to excitatory synapses, and whether N-glycosylation regulates NMDA receptor function.
NMDA receptor transport
Martina Kaniaková, Ph.D.
The goal of this project is to clarify the mechanisms regulating NMDA receptor transport to the cell membrane. We want to identify subunit-specific mechanisms of NMDA receptor transport, and to determine whether NMDA receptor release from the endoplasmic reticulum is dependent on a specific functional state of the receptor. Our results will contribute to a better understanding of the regulation of glutamatergic signaling in health and disease.
Influence of membrane lipids on NMDA receptor function
Miloslav Kořínek, Ph.D.
NMDA receptors are integral membrane proteins whose function can be influenced by the composition of the cytoplasmic membrane. We have discovered that cholesterol, an important structural component of the membrane, has a robust modulatory effect on NMDA receptors. Specifically, cholesterol depletion from the membrane strongly inhibits NMDA receptor function. This is consistent with evidence that cholesterol depletion protects neurons from glutamate excitotoxicity. Our current experiments aim to further characterize the molecular mechanism of NMDA receptor modulation by cholesterol, and to clarify the influence of cholesterol on glutamatergic synaptic transmission. This project will greatly improve our understanding of the role of cholesterol in neurons, particularly as it relates to excitotoxicity.
Synaptic effects and neuroprotective properties of NMDA receptor modulators
Tereza Smejkalová, Ph.D.
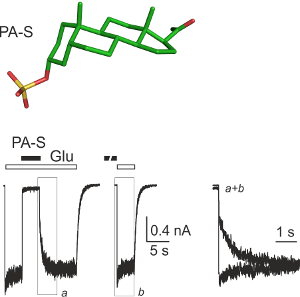
Synaptic activation of NMDA receptors plays a key role in synaptic plasticity and can be neuroprotective. On the other hand, excessive tonic NMDAR activation mediates excitotoxicity. This paradoxical involvement of NMDARs in both neuroprotection and neurotoxicity motivates an interest in pharmacological agents selectively blocking pathological NMDAR signaling while leaving normal synaptic transmission intact. Endogenous neurosteroid pregnanolone sulfate (PA-S) inhibits NMDARs in a use-dependent manner, and at the same time potentiates presynaptic glutamate release. These properties predict that PA-S may selectively inhibit tonically over synaptically activated NMDARs and thus be neuroprotective. We use synaptic electrophysiology to characterize presynaptic and postsynaptic effects of PA-S and PA-S analogues. Using in-vitro excitotoxicity models we aim to identify an optimal combination of steroid synaptic effects for maximal neuroprotection.
Role of linker regions in the signal transduction between the ligand-binding domain and the ion channel of the NMDA receptor
Jan Krůšek, Ph.D.
Given the importance of NMDA receptor function in physiology and pathology, we study the relationship between the receptor structure and its function. We are particularly interested in the role of linker regions connecting the ligand-binding domain and the ion channel. Our results will contribute to a more detailed understanding of the mechanism of ion channel opening and may help identify novel regulators of NMDA receptor function. (This project is in collaboration with Jiří Černý, Ph.D.)
Modulation of NMDA receptors by neurosteroids
Ladislav Vyklický, M.D., DSc.
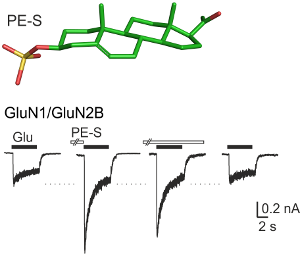
Neurosteroids are steroidal compounds produced by the nervous tissue and thought to regulate neural activity locally through direct modulation of membrane receptors. NMDA receptors, in contrast to AMPA/kainate receptors, are significantly influenced by neurosteroids. Pregnanolone sulfate (PA-S) inhibits NMDA receptors, while structurally similar pregnenolone sulfate (PE-S) has a potentiating effect. We are focusing on the following areas.
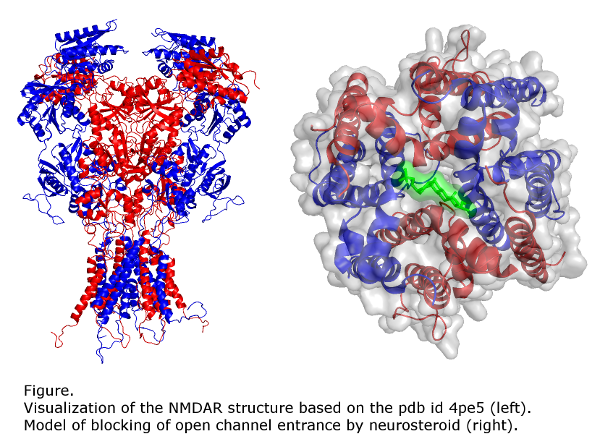
Molecular mechanism of steroid action at NMDA receptors
Our goal is to identify the site(s) and the molecular mechanism(s) of steroid action at the NMDA receptor. Our results suggest that PA-S binds in the extracellular vestibule of the ion channel pore. Kinetic analysis of NMDA receptor inhibition by PA-S and its synthetic analogues suggested that these compounds may inhibit tonically activated NMDA receptors more potently than synaptically activated receptors, a pharmacological profile that would be very useful clinically. Our preliminary results confirm these predictions and suggest that PA-S is neuroprotective without psychomimetic side effects characteristic of other NMDA receptor inhibitors. We are also trying to identify the binding site for neurosteroids with potentiating action at the NMDA receptor.
Role of cytoplasmic membrane in the access and action of steroids at the NMDA receptor
NMDA receptors are transmembrane protein structures and their membrane domains are in direct in contact with and influenced by membrane lipids. Neurosteroids at concentrations active at NMDA receptors form micelles in aqueous solutions. We have developed a model describing the conversion of micellar steroid to its molecular form. Using molecular dynamics simulations we continue to study the interactions of steroids with the membrane.
Relationship between the structure and the function of NMDA receptor-modulating steroids
In collaboration with Eva Kudová, Ph.D. and Hana Chodounská, Ph.D. (IOCB, CAS) we are testing the properties of synthetic analogues of PA-S and PE-S. One of our goals is to identify the structural basis for steroid inhibitory vs. potentiating action at NMDA receptors. In addition, we are searching for highly potent steroids; out of ~200 newly synthesized compounds, several are active at nM concentrations. The most promising compounds are subsequently tested in behavioral assays.
Modeling of NMDA receptors
Trying to complement the above mentioned experimental approaches we study the structure function relationships of NMDA receptors also theoretically. A combination of tools of molecular modeling is employed for understanding the molecular mechanism of opening and modulation of function by interaction with neurosteroids and membrane components. The tools we are using cover the homology modeling, docking of proteins and neurosteroid ligands, molecular dynamics simulation as well as accurate quantum chemical calculations. The computational approach is realized by Jiří Černý, Ph.D. and Michaela Nekardová (Faculty of Mathematics and Physics, Charles University in Prague).