TRP Channels
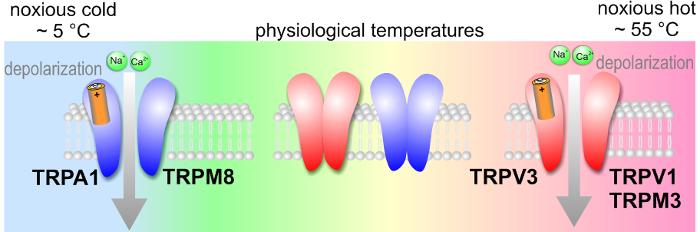
We investigate molecular mechanisms of nociception and thermosensation by focusing on transient receptor potential (TRP) ion channels (Figure). These channels are specifically expressed in primary nociceptive neurones and work in concert to detect potentially damaging stimuli and transduce them into pain signalling. The goal of our research is to understand the physiological significance of a specific subclass of TRP ion channels that are involved in the detection of noxious thermal, mechanical and chemical stimuli through deciphering the molecular and biophysical principles of their operational features that underlie the basis of noxious sensation in mammals.
Research Topics
Viktorie Vlachová, DSc.
Molecular mechanisms of TRPV1 and TRPA1 activation
Vanilloid receptor TRPV1 is an ion channel which acts as a polymodal sensor for multiple noxious stimuli, including temperature, capsaicin, various pungent compounds and acidic solutions. We try to identify molecular determinants and domain interactions that underlie the conformational transitions among resting, open and inactivated (desensitized) state of these channels.
Molecular mechanisms of thermosensation
The ability of temperature sensing is conferred by a group of ion channels from the TRPV (vanilloid), TRPM (melastatin) and TRPA (ankyrin) families that are mainly localized on neurones of nociceptive pathways. We try to clarify the nociceptive mechanisms underlying thermosensation at peripheral nerve endings by focusing especially on TRPV1, TRPV3, TRPM8, and TRPA1.
TRPV1 and TRPA1 as intrinsically voltage-sensitive ion channels
TRPV1 and TRPA1 are intrinsically voltage-sensitive and thermal and chemical stimuli act to increase their voltage sensitivities. We attempt to explore the molecular basis for the voltage-dependent gating in these channels and to understand the physiological significance of this property in nociception.